The frequency of women overdue for cervical cancer screening has increased to 23% in 20191 or over 40 million women.The reasons varied from pain, accessibility issues, lack of information.The National Cancer Institute has recently made a push for improving access to at home screening for HPV through the Last Mile Initiative and the SHIP Trial.
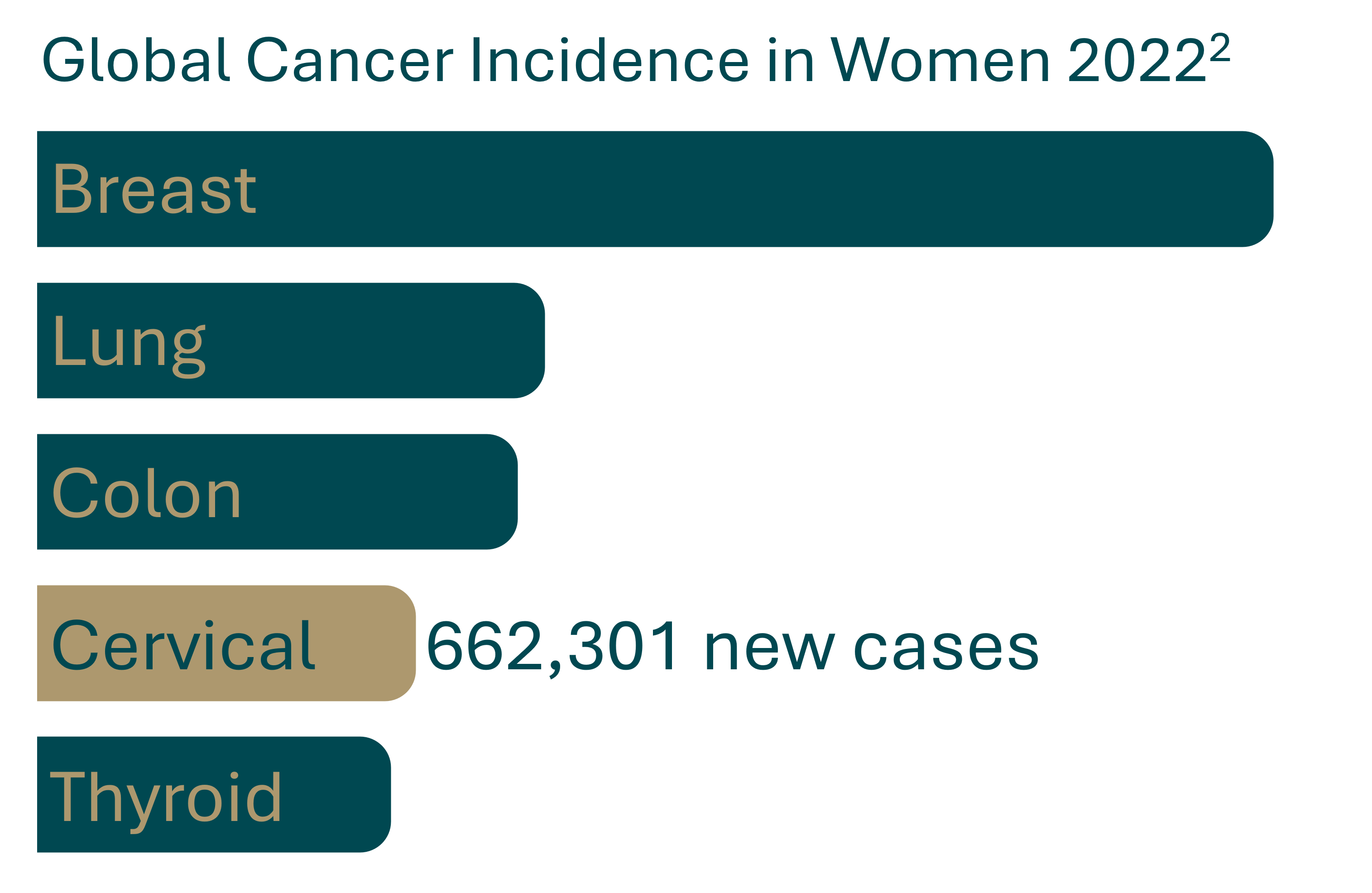
While the vaccine is effective in protection against listed strains, cervical cancer still holds 4th place for most common cancers in women world wide.
.
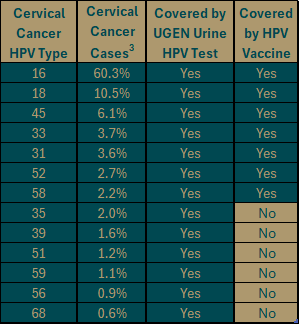
UGEN’s Urine HPV test covers all cervical cancer causing HPV types, including the ones not covered by the vaccine.
Surveys point to anxiety, discomfort, embarrassment, and pain as reasons why patients avoid getting screened4
With a sensitivity of 96%, our kit makes screening for HPV easy, safe, and comfortable using our urine based detection kit we can test for multiple HPV strains right from the patient’s home without having to make an appointment for a pap smear.

Order a test for your patients using our link *HERE*
A collection kit will be mailed to your patient where they follow the instruction and provide their urine that will be placed into our preservation solution.


The sample will have prepaid packaging that will get sent to our expert analysts in San Diego.
The lab will then send the results to you, so that you can continue to make the best healthcare decisions for your patients.

- Suk R, Hong YR, Rajan SS, Xie Z, Zhu Y, Spencer JC. Assessment of US Preventive Services Task Force Guideline-Concordant Cervical Cancer Screening Rates and Reasons for Underscreening by Age, Race and Ethnicity, Sexual Orientation, Rurality, and Insurance, 2005 to 2019. JAMA Netw Open. 2022 Jan 4;5(1):e2143582. doi: 10.1001/jamanetworkopen.2021.43582. PMID: 35040970; PMCID: PMC8767443.
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024 May-Jun;74(3):229-263. doi: 10.3322/caac.21834. Epub 2024 Apr 4. PMID: 38572751.
- Perkins RB, Wentzensen N, Guido RS, Schiffman M. Cervical Cancer Screening: A Review. JAMA. 2023 Aug 8;330(6):547-558. doi: 10.1001/jama.2023.13174. PMID: 37552298.
- Suk R, Hong YR, Rajan SS, Xie Z, Zhu Y, Spencer JC. Assessment of US Preventive Services Task Force Guideline-Concordant Cervical Cancer Screening Rates and Reasons for Underscreening by Age, Race and Ethnicity, Sexual Orientation, Rurality, and Insurance, 2005 to 2019. JAMA Netw Open. 2022 Jan 4;5(1):e2143582. doi: 10.1001/jamanetworkopen.2021.43582. PMID: 35040970; PMCID: PMC8767443.
